To this point in time that did not yet happen. The expectation of the industry before the publishment of the EN ISO 14971:2019 was, that the new standard will also be harmonized with the EU legislation, therefore replacing the previous edition when the Regulations will replace the Directives. Will ISO 14971:2019 be harmonized with the EU legislation? The alignment with the requirements of ISO 13485, EU MDR/IVDR and FDA. The publishment of the new edition is a result of a vote taken by the International Organization for Standardization (ISO) and the International Electrotechnical Commission (IEC) in 2016. 
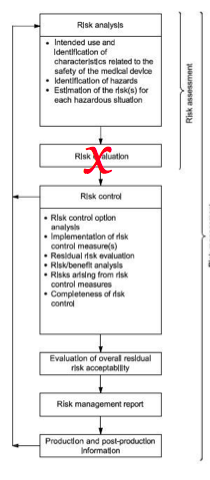
The third edition of the standard for application of risk management to medical devices – the ISO 14971:2019 – was published in December 2019 (along with the EN version), followed by the technical report ISO/TR 24971:2020 in June 2020. This standard establishes the requirements for risk management to determine the safety of a medical device by the manufacturer during the product life cycle. ISO 14971:2019 is a standard for the application of risk management to medical devices.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
